Health, Hormones and Human Performance Part 2
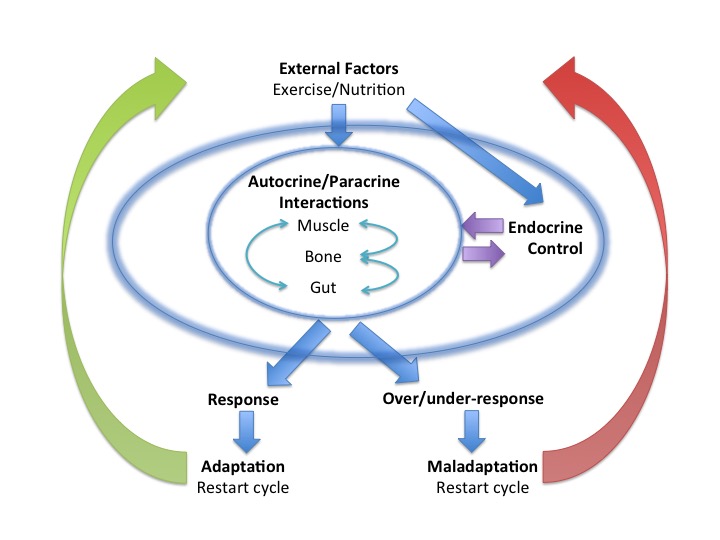
Endocrine and Metabolic aspects of Sports and Exercise Medicine are crucial determinants of health and human performance, from reluctant exerciser through to elite athlete and professional dancer. This is what the recent BASEM spring conference set out to demonstrate. The previous blog described functional disruption of Endocrine networks caused by non-integrated periodisation of the three key lifestyle factors of exercise/training, nutrition and recovery/sleep, can lead to adverse effects on health and athletic performance.

Grace, aesthetic line and ethereal quality belie the athletic prowess required in ballet. What are the Endocrine, metabolic and bone health consequences for this unique group of athletes? Dr Roger Wolman (Medical Advisor to National Institute for Dance Medicine and Science) returned to the important topic of insufficient energy availability in sport/dance where being lightweight confers a performance advantage, resulting in dysfunction in multiple endocrine axes. Dr Wolman discussed his recent research studies in dancers revealing an intriguing synergistic action between oestrogen and vitamin D, which is itself a steroid hormone. Evidence was presented to demonstrate how being replete in vitamin D has beneficial effects on bone, immunity and muscle function. Thus it is key in preventing injury and supporting health in athletes, with particular relevance in premenarchal and postmenopausal women, who are in relative oestrogen deficient states. This presentation will certainly change my clinical practice and, I am sure, that of many in the audience, in ensuring that athletes/patients are vitamin D replete. This may have to be achieved in the form of strategic use of sports informed vitamin D supplementation, given that even walking naked for 5 hours a day outside during UK winter, would not stimulate enough vitamin D production. Therefore, to the relief of many in the audience, Dr Wolman did not recommend this strategy.
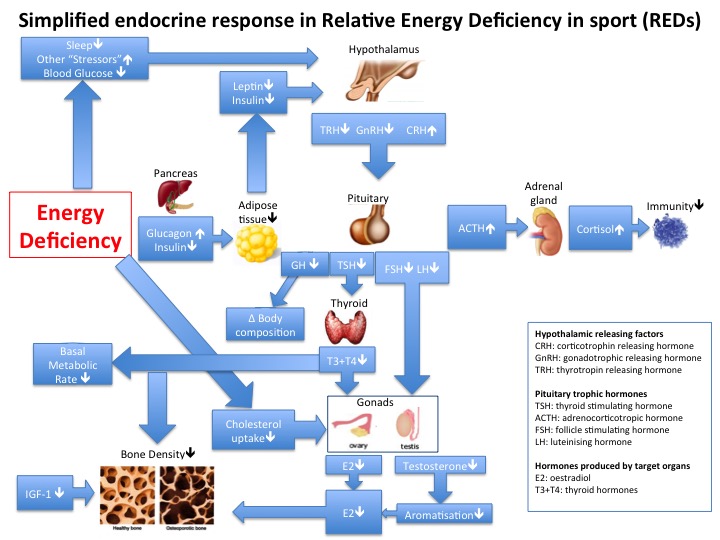
Dr Kate Ackerman (member of RED-S IOC working group) explained why we should all tap into our inner endocrinologist. Sport and Exercise Medicine (SEM) goes far beyond diagnosing and treating injury. Is there any underlying endocrine cause for suboptimal health, performance or injury? Be this an endocrine diagnosis that should not be missed, or a functional endocrine dysfunction due to relative energy deficiency in sports (RED-S). Dr Ackerman explained the importance of the multidisciplinary team in both identifying and supporting an athlete experiencing the consequences of RED-S. New research from Dr Ackerman’s group was presented indicating the effects of RED-S on both health and athletic performance.
Females now have combative roles alongside their male counterparts. What are the implications of this type of intensive exercise training? Dr Julie Greaves (Research Director of the ministerial women in ground close combat research programme) presented insightful research revealing that differences in the geometry of bone in men and women can predispose towards bone stress injury and account for increased incidence in this type of injury in female recruits.
Lunchtime discussion and debate was focused on the determinants of athletic gender, lead by Dr Joanna Harper and Professor Yannis Pitsiladis (International Federation of Sports Medicine). Rather than relying on genetic sex, testosterone concentration was proposed as the criteria for determining whether an athlete competes in male or female events. That testosterone concentration is linked to performance was demonstrated in a study published last year in the BMJ where female athletes in the upper tertile of testosterone were shown to have a performance advantage in certain strength based track and field disciplines. This could potentially be an objective, functional metric used to determine sporting categories for transgender and intersex athletes. The only current uncertainty is how previously high levels of testosterone seen in male, or intersex athletes would have already had an impact on physiology, if this athlete then wished to compete as female and therefore lower testosterone levels with medication.
Nutrition is a key component in optimising health and performance through the Endocrine system. Dr Sophie Killer (English Institute of Sport) explained practical implications for athletes. In a study stimulating a training camp, there were distinct differences between athletes on different regimes of carbohydrate intake in terms of endocrine markers and psychological effects. Those athletes on restricted carbohydrate intake fared worse.
Insulin insensitivity is the underlying pathological process in developing type 2 diabetes mellitus (T2DM) and metabolic syndrome. What is the crucial lifestyle intervention to combat this? Dr Richard Bracken (Swansea University) presented the science behind why and how exercise improves blood glucose control and therefore ultimately risk of developing the macro and microvascular complications of diabetes. T2DM is an increasing health issue in the population, which has to be addressed beyond reaching for the prescription pad for medication. Dr Bracken outlined some effective strategies to encourage the reluctant exerciser to become more active. Having worked myself in NHS diabetic clinics over many years, this was a key presentation at the conference to demonstrate that SEM goes far beyond a relatively small group of elite athletes. Highlighting the crucial role of physical activity in supporting health and performance through optimisation of endocrine networks: uniting the elite athlete and the reluctant exerciser.

Motivate2Move initiative aims to shift the emphasis from treating disease, to preventing disease. Dr Brian Johnson presented the excellent resource for healthcare professionals to encourage, motivate and educate patients in order to consider exercise as an effective and enjoyable way to improve health.
Hormones play a key role in health and human performance, applicable to all levels of exerciser from reluctant exerciser to elite athlete.

References
Health, Hormones and Human Performance BASEM Spring Conference
Video of presentation on Endocrine and Metabolic aspects of Sport and Exercise Medicine from BASEM Spring Conference
Sports Endocrinology – what does it have to do with performance? Keay BJSM 2017
Lifestyle Choices for optimising health: exercise, nutrition, sleep Keay BJSM 2017
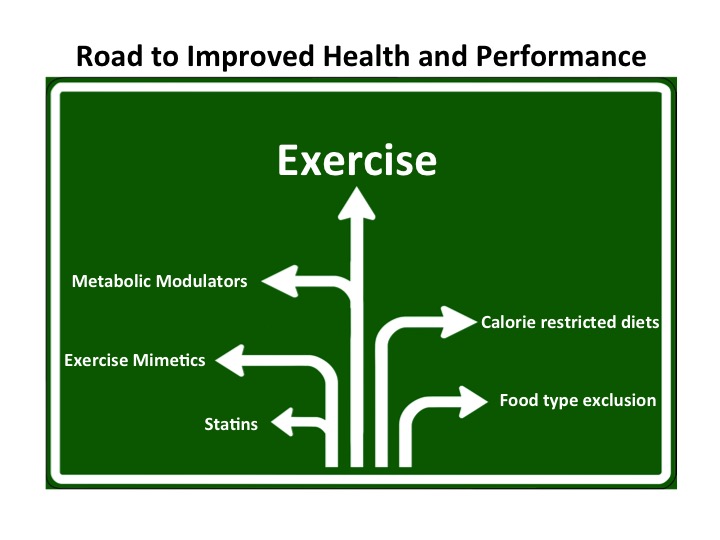
One road to Rome: Exercise Keay, BJSM 2017